It has recently been determined that drug overdoses are now the leading cause of death among Americans under 50 years of age. This finding is just one effect (among many) of the rising U.S. opioid crisis which, as of 2018, has been declared a national public health emergency by the Trump Administration and by the Department of Health and Human Services. With the opioid crisis rising once again to the forefront of U.S. news given the recent Johnson & Johnson opioid lawsuit, learn about the context of the epidemic and recent U.S. countermeasures with HeinOnline.
Before We Get Started:
Don’t miss out! Make sure you have the databases we’ll be mentioning in this post. Follow the links below to start a trial today.
- U.S. Statutes at Large
- U.S. Code
- U.S. Federal Agency Documents, Decisions, and Appeals
- Code of Federal Regulations/Federal Register
- U.S. Federal Legislative History Library
- U.S. Congressional Documents
- GAO Reports and Comptroller Decisions
- World Treaty Library
- Reports of U.S. Presidential Commissions and Other Advisory Bodies – Named to the 2018 List of Best Historical Materials
Understanding Opium and Its Derivatives
A Brief History of Opium Use
Opium is derived from the opium poppy by scoring the immature seed pods and drying the latex which seeps out. Since before the Common Era, the opium poppy and its seeds have been used for food, medicine, and spiritual purposes. Archaeological evidence tells us that the Ancient Greeks consumed opium in a variety of ways, including through poultices, suppositories, and inhalation. Similar opium use appears to have been commonplace throughout other ancient civilizations, including Sumer, Egypt, India, Rome, and Persia.
Opium entered Western medicine as a pain reliever in the 16th century. As a less-harmful alternative to arsenic, mercury, and other contemporary options, the drug became a standard medical treatment for all sorts of illnesses through the 19th century, including dysentery, diarrhea, bronchitis, tuberculosis, psychosis, and female “hysteria.” The pleasure and intense high gained from some forms of opium led to the drug’s recreational use during the same time period.
During the 1800s, purified and synthetic opioids with stronger effects became more popular than naturally-derived opium. Morphine was successfully isolated from opium in 1817, codeine in 1832, and heroin in 1874. In the early 1900s, heroin was introduced commercially as child cough medicine and a non-addictive substitute for morphine.
The early 20th century saw the restriction of opium in many countries with the first international drug control treaty, the International Opium Convention. Signed in 1912 during the First International Opium Conference, the treaty went into force in 1919 intending to impose restrictions on opium exports. The 1961 Single Convention on Narcotic Drugs superseded earlier treaties, prohibiting non-medical use of opium and other controlled substances.
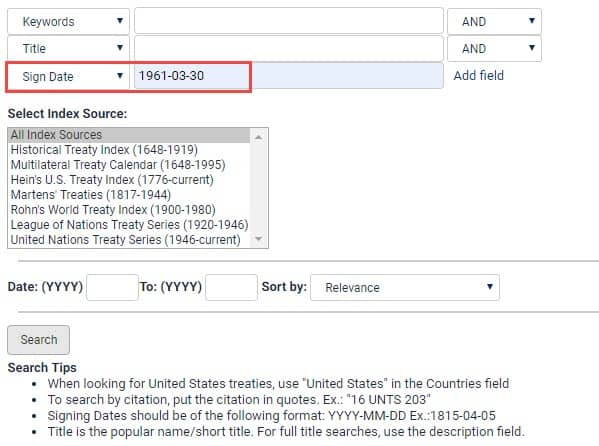
Find these treaties in HeinOnline’s World Treaty Library. In the Treaty Index on the welcome page, change one of the drop-down boxes to “Sign Date” and enter the date in the following format: YYYY-MM-DD. For instance, to find the Single Convention on Narcotic Drugs, enter “1961-03-30” to view the available world treaties signed on that date.
What is an Opioid?
“Opioid” is defined in the most recent edition of the U.S. Code as “any drug having an addiction-forming or addiction-sustaining liability similar to morphine or being capable of conversion into a drug having such addiction-forming or addiction-sustaining liability.” Medically, the term “opioid” refers to a broad class of drugs derived from naturally occurring or synthesized opium. Typically used to relieve or control pain, the most commonly used opioids include those which are legally prescribed—like Vicodin, Percocet, and morphine—as well as illegal drugs such as heroin and illegally obtained fentanyl.
The U.S. Opioid Crisis
The availability and potency of opioids have made them a popular contemporary medical treatment and recreational drug. However, regular use of opioids—whether legally prescribed or illegally obtained—can lead to the development of high tolerance and eventual dependence on the drug. Tolerance and dependence lead to more frequent opioid use and higher doses. While low doses of opioids relieve pain and make the user feel drowsy, higher doses lower the heart rate, slow or stop breathing, and can lead to death. Furthermore, illegally produced drugs—like heroin—can be laced with other opioids, such as the extremely potent fentanyl, to increase their strength. Heroin users unaware of the addition of other drugs are therefore even more likely to suffer a fatal overdose.
The opioid crisis is thus a problem of legal over-prescription and misuse, as well as illegal and unstable recreational use. Over-prescription of opioids in the United States increased during the 1980s and 90s, leading to the United States’ becoming the world’s largest consumer of prescription opioids. Additionally, opioids have become the most commonly prescribed class of medications in the country.
It is unclear from where this pattern of over-prescription stems. Some point to a push by drug companies to expand opioid usage for pain treatment, in response to a late 1990s estimation that a third of the U.S. population experience chronic pain. Others blame the fact that opioids are an inexpensive treatment when compared to other therapies, and that many insurance companies will cover the drugs rather than other treatments.
Regardless, according to the Centers for Disease Control and Prevention (CDC) under the Department of Health and Human Services (HHS), opioid-related deaths by overdose rose from around 8,000 in 1999 to nearly 48,000 in 2017. More than 28,000 of these deaths in 2017 occurred due to fentanyl or fentanyl-related overdose (40% of all drug overdoses that year). In 2017, accidental opioid overdose became one of the most likely causes of death in the United States, surpassing motor vehicle accidents for the first time in history. Finally, as of 2017, drug overdose has become the leading cause of death in Americans under 50.
U.S. Countermeasures in HeinOnline
Past Countermeasures
Back in 1970, President Nixon signed into law the Comprehensive Drug Abuse Prevention and Control Act of 1970. Title II of this act—the Controlled Substances Act—consolidated a number of drug laws to provide a legal foundation for government action against drug and substance abuse. The Controlled Substances Act created five classes of drugs based on their potential for abuse. For example, Schedule I substances have no evident medical use, are considered unsafe, and have a high potential for abuse—it comes as no surprise that as a result, heroin is considered a Schedule I drug. Fentanyl and many other opioids are considered Schedule II drugs under this act, meaning that they have a high potential for abuse and may lead to severe dependence, but also are currently accepted as medical treatment in the United States.
In 2010, President Obama amended the Controlled Substances Act with the Secure and Responsible Drug Disposal Act, allowing individuals, households, and facilities to dispose of their controlled substances to pharmacies in “take-back” programs.
In light of the growing opioid problem, 2016 saw the institution of a number of federal countermeasures to the crisis. Among them, President Obama signed the Comprehensive Addiction and Recovery Act, expanding treatment for opioid addiction and authorizing several million dollars in funding toward opioid research and treatment. Obama also signed into law the 21st Century Cures Act, including $1 billion of state grants toward the fight against the opioid epidemic. View the legislative history of the 21st Century Cures Act in HeinOnline’s Federal Legislative History Library.
Despite this progress, in 2016 Congress also passed the Ensuring Patient Access and Effective Drug Enforcement Act, which requires the Drug Enforcement Administration (DEA) under the Department of Justice to determine “imminent danger to the public health and safety” before suspending the controlled substances privileges of manufacturers and distributors, decreasing the DEA’s ability to intervene in the opioid crisis.
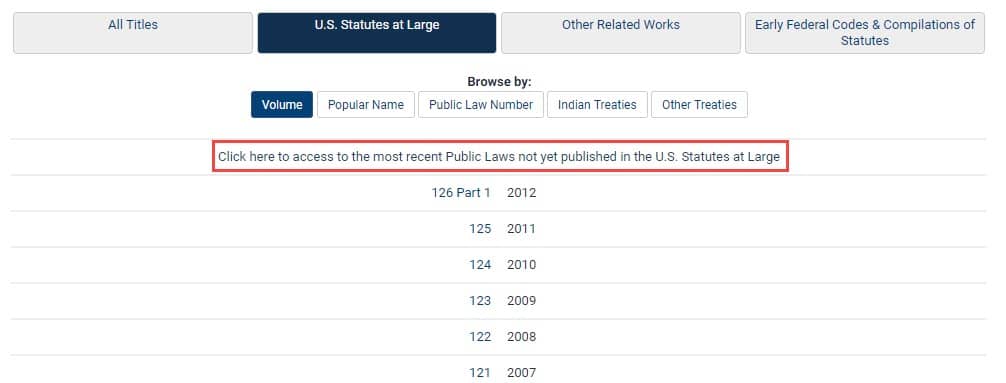
Though the more recent acts have yet to be officially published in bound volumes by the Government Publishing Office (GPO), users can still find the acts in HeinOnline’s U.S. Statutes at Large. From the home page, simply click on the below link to access the most recent public laws that have not yet been published.
The Current Administration
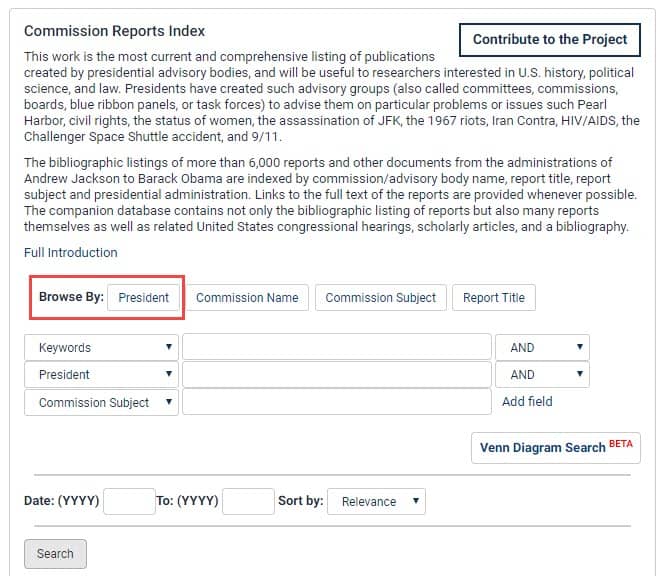
In 2017, President Donald Trump established a commission to combat the opioid epidemic with Executive Order 13784, headed by Governor Chris Christie. View the final report of the President’s Commission on Combating Drug Addiction and the Opioid Crisis in HeinOnline’s Reports of U.S. Presidential Commissions and Other Advisory Bodies database. The welcome page of the database hosts a Commissions Report Index. Within the index, click “Browse by President” and select Trump’s name to view a list of his commissions, councils, task forces, and more—including the Commission on Combating Drug Addiction. Users can also browse by commission name, subject, report title, or they can search the metadata using the drop-down boxes below.
In April of 2017, the Department of Health and Human Services announced a five-point strategy to combat the opioid crisis. The aims include:
- Better prevention, treatment, and recovery services
- Better data on the epidemic
- Better pain management
- Better targeting of overdose-reversing drugs
- Better research on pain and addiction
In August of the same year, agreeing with the report recommendations from his commission, Trump declared the opioid crisis a national emergency. In October, Trump ordered the Acting Secretary of the Department of Health to declare a public health emergency. View the report on this decision from the Government Accountability Office in HeinOnline’s GAO Reports and Comptroller General Decisions.
The past year has seen advancement in fighting the opioid crisis, as well. In June of 2018, President Trump announced his Initiative to Stop Opioid Abuse and Reduce Drug Supply and Demand. In October of the same year, Trump signed into law the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (also known as the SUPPORT for Patients and Communities Act). The act sought to increase availability for treating, preventing, and recovering from opioid addiction, as well as crack down on the illicit use of drugs such as fentanyl. Though the act has not yet been published by the GPO, view the CRS report for the act in HeinOnline’s U.S. Congressional Documents. In addition, users can find President Trump’s statement on the passage of the act in HeinOnline’s Federal Register.
As the U.S. continues to tackle its opioid crisis, stay in the know with HeinOnline. Keep up with blog posts on current events and hot topics by clicking the Subscribe button at the top of this post.
Don’t forget to connect with HeinOnline on our social media platforms: Facebook, Twitter, Instagram and YouTube.



